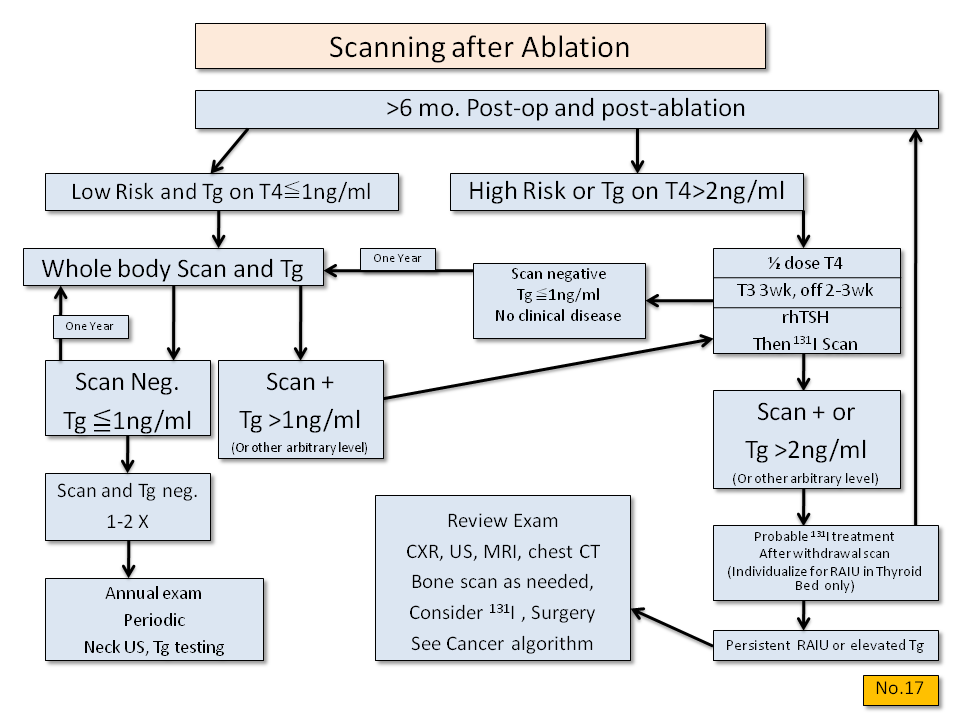
Thyrogen has been approved for use in preparation for 131-I scanning and TG testing. Avaliable data indicate it is nearly (93%) as effective as a standard hormone withdrawal preparation for detecting residual thyroid tissue (benign or malignant) by scan and TG assay. The protocol suggested for use has been developed using a standard dose of Thyrogen (0.9mg) given on days one and two, dosing on the third day with 4 mCi of 131-I, scanning and TG testing on the fifth day, and measuring TG in a reliable assay sensistive to <1 ng/ml. Use of Thyrogen for 131-I therapy is logical but not yet established. The therapist will need to individually evaluate risk, TG values, and scan results in relation to the patient's age, tumor stage, and prior therapy. The role of 131-I therapy in patients with negative scans and elevated TG is uncertain.
